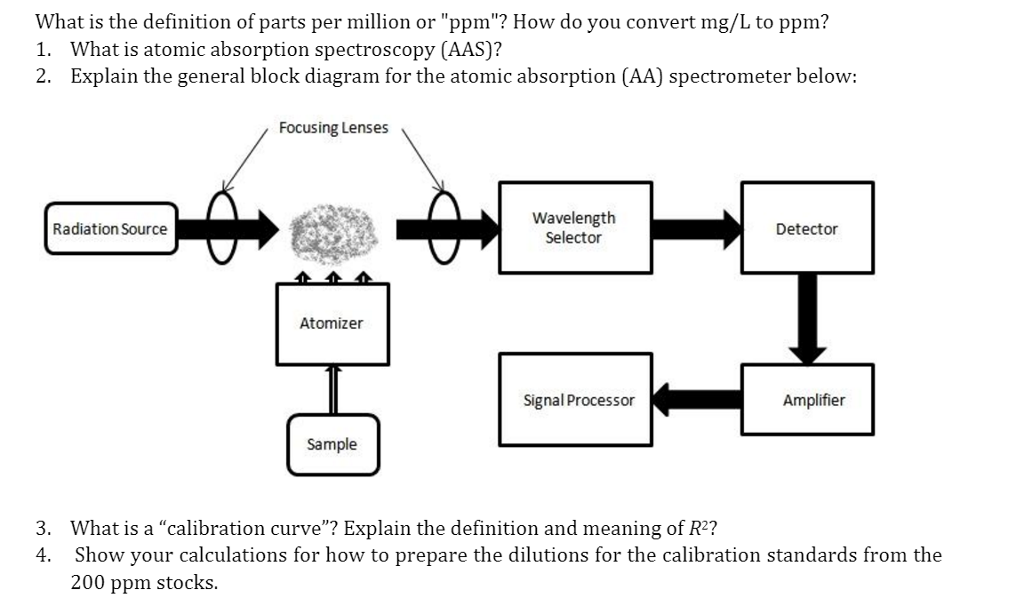
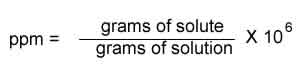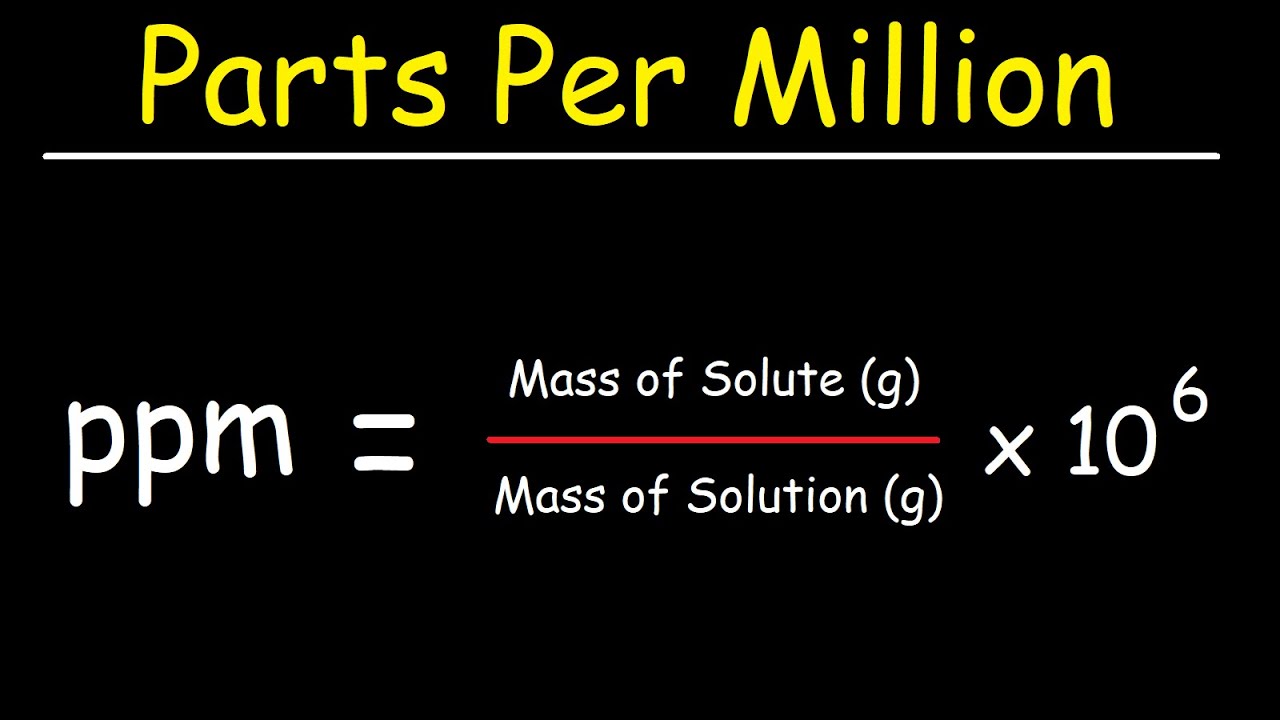What is PPM?| Parts Per Million Calculation | Quality Rejection| NC Product | Explained with example - YouTube

SOLVED: Percent by definition is parts per hundred: % (wlw) (mass of solutelmass of solution) x 100. For very dilute solutions, chemists will often use parts per thousand or parts per million (

Parts per Million Concentration. Parts per Million (ppm) is the ratio of the number of grams of solute for every one million grams of solution. parts. - ppt download